



Kategorien:Peptid-Fertigprodukt, Peptide und ihre Dosierungen
Freies (1) 30 ml bakteriostatisches Wasser
mit qualifizierten Bestellungen über$ 500 USD.
(Ausgeschlossene Kapselprodukte, kosmetische Peptide, Promo -Codes und Versand)
Tesamorelinist ein wachstumshormon-freisetzendes Hormon (GHRH) -Analogon, das klinisch für die Behandlung der HIV-assoziierten Lipodystrophie (dysfunktionale Fettabscheidung) verwendet wird. Es wird auch nach seiner Fähigkeit erforscht, die Gesundheit der peripheren Nerven zu verbessern, das Fortschreiten einer leichten kognitiven Beeinträchtigung und der Reduktion der Fettmasse zu verlangsamen.
Produktnutzung:Dieses Produkt ist nur als Forschungschemikalie gedacht.Diese Bezeichnung ermöglicht die Verwendung von Forschungschemikalien ausschließlich für In -vitro -Tests und Laborversuche. Alle auf dieser Website verfügbaren Produktinformationen dienen nur zu Bildungszwecken. Die körperliche Einführung jeglicher Art in Menschen oder Tiere ist gesetzlich streng verboten. Dieses Produkt sollte nur von lizenzierten, qualifizierten Fachleuten behandelt werden. Dieses Produkt ist keine Droge, Nahrung oder Kosmetik und darf als Medikament, Nahrung oder Kosmetik nicht missbraucht, missbraucht oder missbräuchlich werden.
Tesamorelin ist ein Wachstumshormon-Freisetzungshormon (GHRH) -Analogon, das aus Standard-GHRH besteht, zu dem eine zusätzliche trans-3-Hexansäure-Gruppe zugesetzt wurde. Tesamorelin wurde von Theratechnologies of Canada produziert und wurde 2010 zum neuesten Medikament, das von der FDA zur Verwendung in HIV-assoziiertem Lipodystrophie zugelassen wurde. Das Peptid wurde auch wegen seiner Fähigkeit zur Verbesserung der peripheren Nervenregeneration und als potenzielle Intervention für milde kognitive Beeinträchtigung (MCI), die Vorausdementierung, und die Vorausdementien untersucht.
Als aGHRHAnalog, Teamorelin hat alle die gleichen Effekte wie GHRH- und GHRH -Analoga wiePERMORELIN, GRF (1-29), CJC-1295usw. Die Zugabe von Trans-3-Hexansäure zu Teamorelin macht es in menschlichem Plasma stabiler und erhöht somit seine Halbwertszeit. Trotz dieser Erhöhung der Halbwertszeit bewahrt Tesamorelin wie CJC-1295 die physiologische Wirkung von GHRH und weist somit weniger Nebenwirkungen auf als ähnliche Moleküle, die das normale pulsatile Wachstumshormon (GH) Freisetzung ausklopfen.
Die primäre Verwendung für Teamorelin ist die Behandlung der HIV-assoziierten Lipodystrophie, die sowohl als Folge einer HIV-Infektion als auch als Nebenwirkung der antiretroviralen Therapie zurückzuführen ist. In der Lipodystrophie akkumuliert sich Fett sowohl im Bauch als auch in anderen Körperbereichen übermäßig. Der dafür verantwortliche physiologische Mechanismus ist nicht klar verstanden, aber es wird angenommen, dass häufig verwendete Protease -Inhibitoren eine große Rolle bei der Pathogenese der Lipodystrophie spielen[1].
Patients suffering from lipodystrophy initially had diet, exercise, and a handful of ineffective medications to rely on for treatment. If those did not work, surgery was a last-ditch, often ineffective, and frequently complicated solution. In 2010, however, the FDA approved tesamorelin specifically for the treatment of HIV-associated lipodystrophy. The drug has been found to reduce adiposity by nearly 20% in this population [1]. Research suggests that tesamorelin is approximately 4 times more effective in reducing adiposity than all of the other available therapies combined [2].
People with HIV are at increased risk of developing cardiovascular disease (CVD), in part due to abnormal fat deposition and in part due to the actions of antiretroviral drugs themselves. Prevention of CVD in HIV-positive individuals is considered to be the most important medical intervention for long-term well-being, after highly active antiretroviral therapy (HAART) of course. Until recently, statins have been the cornerstone of medical management in this population.
Research shows that tesamorelin, in addition to decreasing lipodystrophy, also reduces triglyceride levels, total cholesterol levels, and non-HDL-C levels in HIV-positive patients. A 15% reduction in visceral adipose tissue by tesamorelin correlates with a 50 mg decrease in trigylceride levels[3], [4].
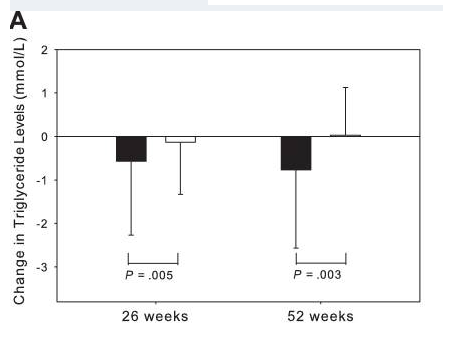
It is worth noting that ectopic fat deposition, as seen in lipodystrophy, is associated with inflammation. Inflammation of any kind is a risk factor for CVD. Visceral adipose tissue, liver fat, and epicardial fat are all independently associated with increased risk of CVD. By reducing ectopic fat deposition, tesamorelin directly decreases inflammation and an individual’s risk for CVD.
Recent evidence suggests that HAART is associated with a number of endocrine and metabolic problems, including growth hormone (GH) deficiency. It appears that the pituitary gland is altered in HIV infection and, as a consequence, approximately one third of patients with HIV who are taking HAART have GH deficiency[5]. This may, to some extent, explain why lipodystrophy is so common in individuals with HIV and also why tesamorelin is such an effective treatment. Tesamorelin is a safer and more effective way to raise GH levels than administration of exogenous GH, particularly in HIV-positive individuals.
Peripheral nerve damage can be a consequence of injury, diabetes, or even surgical interventions. It often results in debilitating problems with both motor and sensory function in the affected area, but there is little that can be done to correct the problem because nerve cells are notoriously difficult to regenerate. Research, however, suggests that therapies based on growth hormone manipulation may improve peripheral nerve injury and increase both rate and extent of healing[6]. Tesamorelin is currently the leading candidate for such intervention, in part because it already has FDA approval.
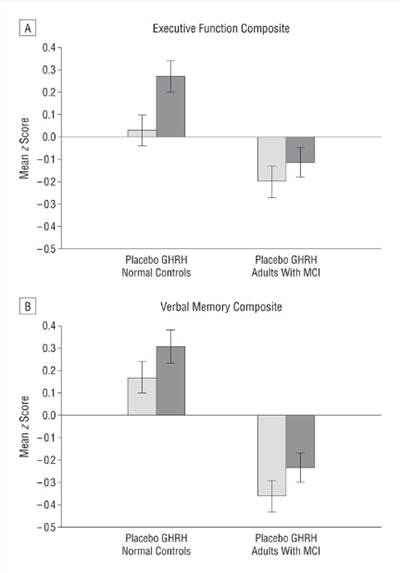
There is now evidence to suggest that GHRH analogues, like tesamorelin, are effective in enhancing cognition in patients suffering from the early stages of dementia. A large, randomized, double-blind, placebo-controlled study at the University of Washington School of Medicine, carried out over twenty weeks, suggests that tesamorelin and other GHRH analogues may impact dementia by increase gamma-aminobutyric acid (GABA) levels in the brain and by decreasing myo-insoitol (MI) levels[7]. These findings open up a pathway for using tesamorelin in the treatment of dementia, but also suggest new areas for scientists to explore as they look for a cure or a preventative.
Because it is FDA approved for use in humans, tesamorelin is an attractive peptide for ongoing clinical research. It is currently under review for its ability to reduce cardiovascular disease in HIV, improve healing of peripheral nerves following injury, and slow the progression of dementia. Clinical trials are already underway in several different areas.
Tesamorelin exhibits minimal side effects, low oral and excellent subcutaneous bioavailability in mice. Per kg dosage in mice does not scale to humans. Tesamorelin for sale at
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.
PeptideGurus is a leading supplier of American-made research peptides, offering top-quality products at competitive prices. With a focus on excellence and customer service, they ensure a secure and convenient ordering process with global shipping.
CONTACT